by Analia Katriniuk | Sep 23, 2025 | Blog
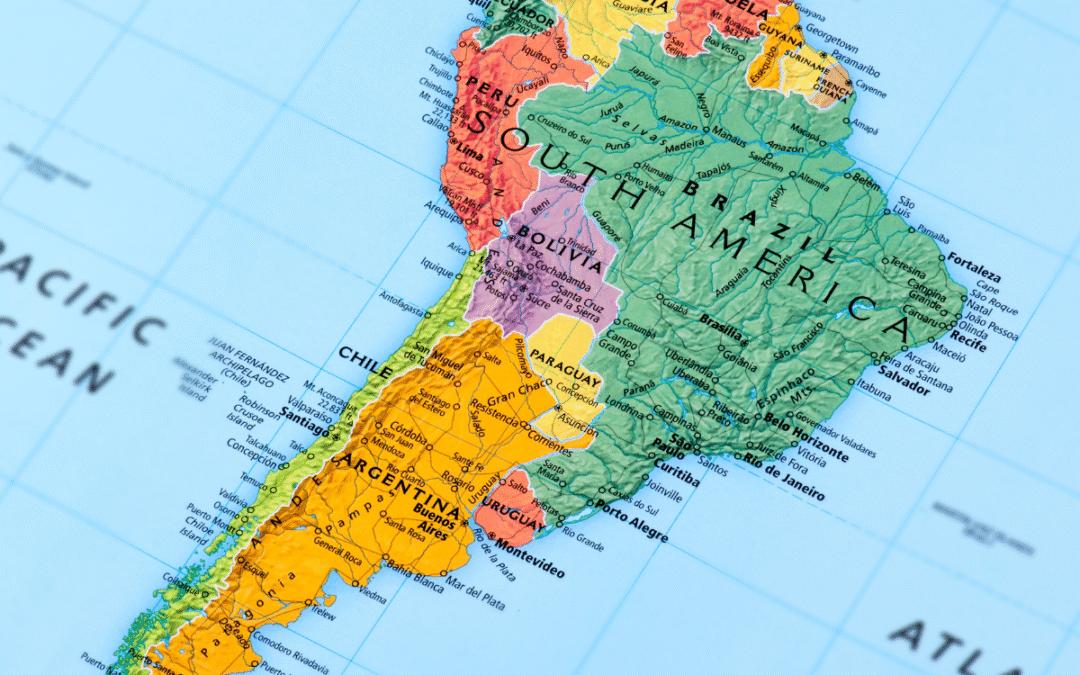
Expanding into Latin America offers tremendous opportunities for businesses, particularly in sectors such as medical devices, pharmaceuticals, cosmetics, and consumer goods. However, navigating regulatory compliance in the region can be complex due to varying laws,...

by Analia Katriniuk | Sep 17, 2025 | Blog
On September 15, 2025, the Instituto de Salud Pública de Chile (ISP) announced the implementation of the World Health Organization’s (WHO) international pharmacovigilance standard to enhance the safety monitoring of medicines and vaccines in Chile. 🔍 What Is the WHO...

by Analia Katriniuk | Aug 14, 2025 | Blog
After moving into our new headquarters in Buenos Aires last year, LAMA took a major step forward by applying to ANMAT for authorization to act as local representatives and importers for medical devices. We are delighted to officially announce that this permission has...

by Analia Katriniuk | Aug 12, 2025 | Blog
Recently, LAMA announced the launch of our expanded post-market surveillance offering, including services in pharmacovigilance, technovigilance, and cosmetovigilance. These disciplines work to ensure that medicines, medical devices, and cosmetics remain safe,...

by Analia Katriniuk | Jul 23, 2025 | Blog
Earlier this week, ANMAT, Argentina’s national regulatory authority, prohibited the sale of Colgate Total Active Prevention Clean Mint toothpaste due to safety concerns linked to this specific presentation of the product. While the move made headlines, largely due to...

by Analia Katriniuk | Jul 16, 2025 | Blog, Uncategorized
Following the recent announcement of our expanded post-market surveillance services at LAMA, we’re now taking a closer look at one of the most critical components of that offering: pharmacovigilance. While post-market surveillance covers a wide range of activities,...